A plaque on the side of the Saint Mary’s Hospital in Paddington, London, UK, commemorates the discovery of penicillin, the first antibiotic, in 1928. Fungal spores, blown by the wind, landed on Sir Alexander Fleming's Petri dishes killing the bacteria he was growing leading to a revolution in medicine.
Doctors, Antimicrobial Resistance and the Fleming Fund
The Fleming Fund invests substantially in improving hospital laboratories. A vital part of this is ensuring that laboratories are used properly by doctors. Doctors are responsible for requesting laboratory tests on patient samples, collecting clinical data (e.g. by completing request forms and making good clinical notes) and are the end-users for individual laboratory results to inform patient care and antimicrobial treatment. Also, in most hospitals and many community healthcare settings, antimicrobial prescribing decisions and guidelines are made by doctors.
Because doctors are the primary prescribers and test results from samples are the foundation of AMR surveillance data, doctors are critical to improving surveillance data and antimicrobial use.
However, engaging and communicating with doctors can be challenging. Many doctors are accustomed to prescribing long courses of broad-spectrum antimicrobials as a precaution, particularly if reliable bacteriology laboratories are unavailable. Similarly, doctors may not fully appreciate the role of laboratories when they already have the authority to prescribe antimicrobials. It can be difficult to change these behaviours.
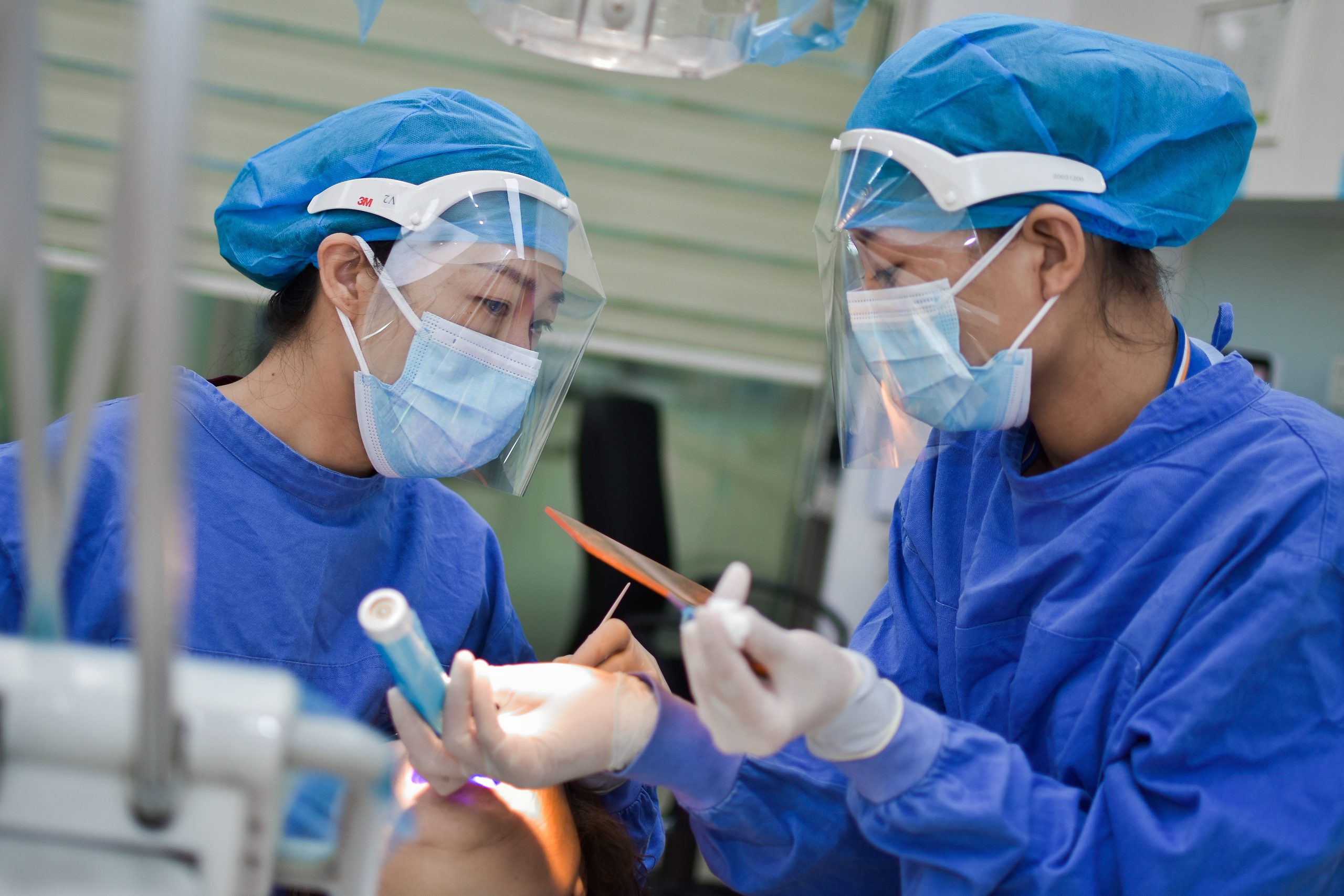
Studies have shown that simply informing doctors, for example by providing guidelines or a single workshop, is the least effective method of achieving behaviour change. Doctors have spent many years in undergraduate and professional training and are strongly influenced by medical hierarchies. A junior doctor is more likely to adhere to instructions from senior consultants, rather than follow new guidelines or advice from other junior doctors, pharmacists, nurses or workshop facilitators.
Instead of just informing, there needs to be ongoing collaboration and empowerment of clinicians, with doctors included in all aspects of the programme to ensure they have a sense of ownership of the efforts to control and contain AMR.
The challenge of clinical engagement was discussed at the recent Fleming Fund Delivery Partner Event. Below are some suggestions from the discussions for how grantees can promote involvement of doctors across the programme, encouraging them to affect policy and regulatory change, take samples and improve prescribing practices.
Tips for Clinical Engagement:
- Involve clinical microbiologists. Some countries have clinical microbiologists who have trained as medical doctors and then specialised at post graduate level in laboratory medicine. Look for national associations of clinical microbiologists and partner with them or recruit a clinical microbiologist for the country grant programme.
- Involve other medical bodies such as professional associations or Colleges of Physicians / Surgeons.
- Advocate for the inclusion of front-line clinicians in AMR programmes and governance systems. Look for champions who can generate activism within the profession within national AMR committees and on hospital boards.
- Promote inter-disciplinary working and learning through Antimicrobial Stewardship Committees/Site Surveillance Committees. Ensure that senior clinicians are involved in AMS/site surveillance committees. Hold regular infection meetings and invite all doctors, nurses, pharmacists and laboratory staff to discuss interesting cases, local resistance data, antimicrobial use and hospital acquired infections.
- Develop hospital induction training programmes on AMR for all new staff including doctors, nurses and paraclinical staff, tailored to their specific duties.
- Involve clinicians in developing the laboratories. For example, involve doctors in drafting sampling guidelines and request forms, to improve their understanding of laboratory processes and turnaround times.
- Make sure that laboratory quality management systems include clinical components such as reporting times for critical results (e.g. Gram stain results) and sample request form completeness.
- Consider how to improve communication between laboratory staff and doctors, rather than just relying on IT systems. Can the laboratory telephone crucial results or send them by WhatsApp? Do laboratory staff contribute to teaching / training sessions? Are laboratory staff involved in relevant clinical meetings? Do laboratory staff receive any feedback on interesting cases or results?
At the site level, encouraging doctors to use laboratories is essential to improve data collection, clinical practice and treatment. At the national level, doctors need to be at the forefront of efforts to contain and control the effects of antimicrobial resistance. By involving doctors across the programme there is a greater prospect of improvement and sustainability.
Sources:
Rabkin SW et al, 2019. Physician Engagement: the Vancouver Medical Staff Association engagement charter; Clinical Medicine 19:278-81 Shah N et al, 2015. Towards changing healthcare workers’ behaviours: a qualitative study exploring non-compliance through appraisals of infection prevention and control practices. Journal of Hospital Infection 9:126-134
More Like This
The UK government's Fleming Fund project in Pakistan, led by grantee DAI and managed by Mott MacDonald, won the global antimicrobial stewardship prize from the AMR Industry Alliance for bringing antimicrobial resistance (AMR) awareness to the forefront of clinical care.