Supported by a range of key stakeholders (including the Fleming Fund), the World Health Organization (WHO) recently published the [Third Progress Analysis](https://apps.who.int/iris/handle/10665/361822) of the Implementation of Antimicrobial Resistance National Action Plans (NAP-AMRs) in South-East Asia.
NEW: Logistical Challenges and Solutions to Building Surveillance Systems report published
As part of its laboratory strengthening activities, the Fleming Fund partners with the International Procurement Agency (IPA), to help it procure and deliver highly specialised laboratory equipment around the world.

The instruments and consumables that allow technicians to effectively identify bacteria, conduct antimicrobial susceptibility testing and culture patient samples can cost upwards of £100,000 and are manufactured by only a few specialist companies globally. Additionally, many of the instruments and consumables have a short shelf-life, are delicate, sensitive to transport and can require a cold chain.
These factors, often coupled with limited infrastructure in receiving countries and challenging import taxes and restrictions, further complicate the delivery of critical equipment.
As a result, the Fleming Fund asked IPA to deliver a report, analysing logistical and procurement challenges and delivering recommendations for improved efficiency and transport times.
The report includes a country by country analysis of the challenges and constraints to delivery of laboratory equipment across Asia and Africa.
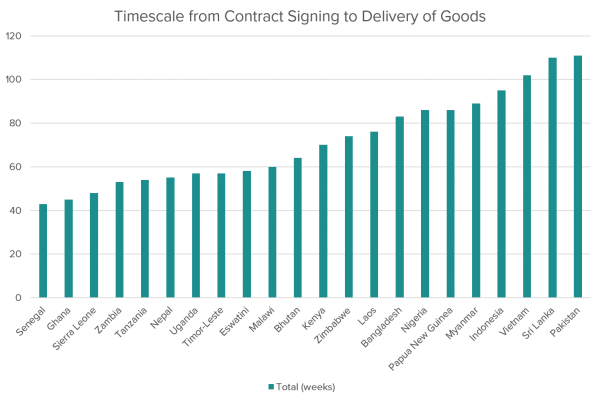
Findings from the report noted that “centralised procurement” (through the Management Agent and IPA) of specialised instruments reduced costs and saved time, but that overall political stability and civil service maturity were critical to facilitating quick the import of items.
Recommendations to improve delivery and transport included:
- Deciding early if tax exemptions are needed. Import levies on goods are common across the world and while donated goods are usually exempt from import taxes, obtaining exemptions can be time-consuming and potentially problematic. Import duties do support local economies and infrastructure, so claiming the money back can be counterproductive to development aims. IPA recommends deciding on whether import tax exemptions will be needed early on and then partnering with exempted local organisations to facilitate a quick customs release, where needed.
- Using local partners. Selecting local partners with medical procurement expertise and familiarity with national procurement processes is important. Local partners are critical to obtaining necessary import licenses which is usually one of the main reasons for supply chain delays. Strengthening local supply chains is an important component of longer-term sustainability.
- Support countries to develop planning & forecasting systems. Many countries operate with “output led” procurement systems, meaning items are ordered as needed with limited forecasting/future planning involved. For laboratory stock systems and equipment deliveries to function effectively, many government logistics specialists need more training on planning and procurement methods. Additionally, countries should be encouraged to develop framework agreements, meaning specific suppliers are “pre-qualified” by the government. Framework agreements save time and reduce lengthy tender processes.
The full report and further country level analysis is available below.
The full document is available here
View pdf (1.38 MB)More Like This
Studies & Reports
From Studies & Reports, 11 countries in South-East Asia are making progress against their AMR National Action Plans, new WHO report shows , Date: 05/01/2023
Studies & Reports
From Studies & Reports, OIE release their Third Annual Report on Antimicrobial Agents intended for use in Animals , Date: 14/02/2019
The Third Annual Report on Antimicrobial Agents intended for use in Animals is now published and includes data from 155 countries.