Antimicrobial resistance (AMR) data is vital for informing local interventions, enabling hospitals to improve infection control and treatment strategies. With Fleming Fund Country Grant investment, the Robotic Process Automation 'bot' technology was introduced − now operational in 23 out of 26 hospitals − to support Nepal’s AMR national surveillance network.
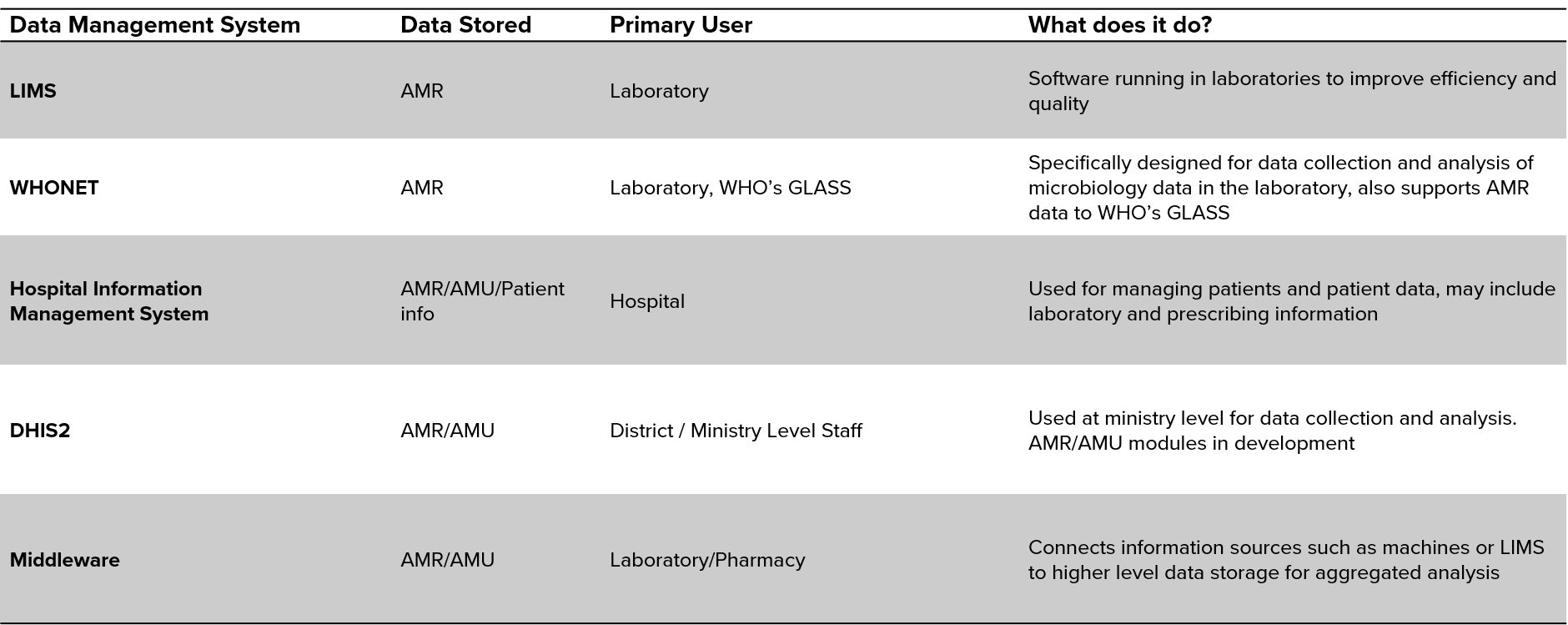
Tools of the Trade: Data Management Software in AMR Surveillance
Robust information management systems are essential for managing and analysing large volumes of data from AMR surveillance systems. In animal and human health, data must be quality assured, communicated from laboratories to clinicians, veterinarians, patients or farmers, and sent to national data coordinating centres. Data must also be interrogated at the regional, national and international level to explore AMR patterns and trends.

Laboratory Information Management Systems ▾
Most laboratories use a Laboratory Information Management System (LIMS). LIMS can be used in real time for sample flow management, automatic entry of laboratory results, quality management (e.g. to monitor reporting times) or to flag unexpected results. Ideally, a LIMS should also be able to communicate with hospital and patient information systems so that laboratory results can be linked directly with additional patient data such as admission date and outcome data.
Most laboratories have a single LIMS for all types of samples. Often, these have been designed for haematology and biochemistry and are not ideal for bacteriology, where workflows are more complex (a single sample may need to be dealt with over several days) and there may be several results for a single sample (positive or negative, identification of organism or organisms, multiple AST results and additional testing such as minimum inhibitory concentration (MIC)). Importantly, many LIMS are not able to store raw data from susceptibility tests, such as zone diameters or MIC values, and can only store the interpretations – resistant, intermediate or susceptible. As break-points and guidelines are regularly updated, this makes it difficult to compare data over time and look for trends.
A key function of the Fleming Fund Country Grants is to work with supported sites to ensure they have a functioning LIMS. Country Grantees are also tasked with ensuring systems are in place which can aggregate and report data to national surveillance systems and GLASS.
WHONET ▾
One option for laboratories is WHONET, a free software designed for entering and analysing data from microbiology laboratories from the World Health Organization. WHONET does not provide all the sample tracking functions of a typical LIMS, however it can be used for basic data entry and analysis and is a good standalone option if the laboratory does not have a large sample throughput to justify purchase of a full LIMS. Alternatively, where laboratories have an existing system, it can usually be configured to export data to WHONET, allowing sites streamline GLASS reporting.
WHONET software can be easily installed on an ordinary PC, can accept and deliver data from a LIMS, and can receive data from automated analysers. It can be used for veterinary, food and environmental samples as well as those from humans.
Regular free updates provide it with the latest breakpoints from the European Committee on Antimicrobial Susceptibility Testing (EUCAST) and Clinical and Laboratory Standards Institute (CLSI) and it can also store the zone diameter and MIC values in case retrospective analysis is needed. Because it is designed specifically for microbiology, it has many practical features to aid entry of isolate data. These include “expert rules” so that important or unusual results are flagged.
Hospital Information Management Systems ▾
In addition to LIMS, some hospitals have IT systems to help manage patient data. These can vary from basic software to record admissions and discharges, to complete patient management systems which integrate patient demographic data, laboratory and radiology results, and prescribing information. Ideally, a LIMS should be linked with the hospital information system, so that laboratory results can be paired with demographic, clinical or treatment data.
National Health Information Management Systems ▾
At the national level, health information management systems are used to aggregate and analyse surveillance data from clinical practice, which sometimes includes data from laboratories. An example is the District Health Information System 2 (DHIS2). This is an open source software platform which can be used to record, visualise, analyse and share district level health information. It can be used in different sectors such as agriculture and water and adapted to suit specific national data needs. It is currently used at the national/ministerial level in nearly 60 countries to analyse all country-level health data, develop routine health reports, understand staffing and equipment needs and monitor trends and outbreaks.
DHIS2 can be useful at a national level for aggregating all AMR/AMU data for the whole country into a single place and providing regular reports and dashboards monitoring key indicators. DHIS2 is not a LIMS and cannot be used for laboratory results currently, so the possibilities of integrating DHIS2 for antibiotic surveillance are beginning to be explored for both AMR and AMU data. This requires development of “modules” that can be added into central DHIS2.
Conclusion
Each software collects different types of data at different levels. Ideally, it should be possible to integrate all data into a single platform, so that trends and outbreaks can be tracked at different levels. For example, comparing AMU data with information on outbreaks might clarify why there has been a change in drug use in a particular area.
To do this, additional “middleware” software may be needed that can take data from lots of different platforms (e.g. a LIMS or WHONET) and bring it together in one place (for example in DHIS2). The immediate need for this will vary from country to country: in many cases, there is very little data at present and the focus of the country grant should therefore be on ensuring good quality data is being collected.
More Like This
Stories
From Stories, Transforming AMR surveillance in Nepal through data automation , Date: 30/04/2025
Since 2019, Fleming Fund grantee, International Vaccine Institute (IVI), has led the CAPTURA consortium to expand the volume of historical data for antimicrobial resistance (AMR), consumption (AMC), and use (AMU) across 12 countries in South and Southeast Asia.